Frequently Asked Questions
- All
- AAS
- BOD
- COD
- Colour
- Conductivity
- Cyroscope
- Density
- Flame Photometry
- General Laboratory
- ICP
- Industry Specific
- Organic Standards
- pH
- Pharmacopoeia
- Physicochemical
- Redox
- Spectrophotometry
- TDS
- Temperature
- Titration
- TOC
- TSS
- Turbidity
- Viscosity
- Water
Turbidity Standards Continuous Improvement Initiative
We wanted to take this opportunity to inform you that Reagecon will be updating the number of significant figures for the Test Results on the Certificates of Analysis (CoA) for our range of Turbidity Standards.
The change is part of a continuous improvement initiative at Reagecon and provides you, the customer, with even greater confidence in the Turbidity results stated on the CoA. The change also ensures that all users of Reagecon’s Turbidity Standards are in adherence with the European Pharmacopeia resolution criteria.
Further information can be viewed here
Cryoscope Standards (NIST)
Reagecon’s Cryoscope Standards are traceable to the National Institute of Standards and Technology (NIST) and are manufactured and certified for use on all Cryoscopes compliant to International Reference Standard ISO5764 / IDF108 “Milk – Determination of freezing point – Thermistor cryoscope method.”
Our Cyroscope Statement can be viewed here
In-Vitro Diagnostic Devices – IVD Statement
Products which are manufactured by Reagecon, are not In-Vitro Diagnostic Devices. These products are not intended for parenteral or in-vivo use of any type. It is imperative that our products are not to be used in vitro.
However, our products can be used for general laboratory use or for external quality assessment as per the exemptions in the IVDR Regulations.
Our IVD Statement can be viewed here
Turbiform™ Stabilised Formazin Turbidity Standards – Storage and Handling Instruction Sheets
As part of a continuous improvement initiative, Reagecon has taken the decision to move away from providing paper copies of the “Storage and Handling Instruction Sheets” for our Turbiform™ Stabilised Formazin Turbidity Standards.
The “Storage and Handling Instruction Sheets” for our Turbiform™ Stabilised Formazin Turbidity Standards are now available via 2-D Barcodes on the Product Labels.
This change will reduce the amount of paper consumption by both Reagecon and our customers and it will make the production processes for Turbiform™ Stabilised Formazin Turbidity Standards more efficient See More …
Temperature vs pH Value for DIN Buffer Standards
Reagecon’s pH Buffer Solutions / Standards have their pH value certified at a single temperature.
To facilitate use of our pH Buffer Solutions / Standards, Reagecon include a table of typical pH values at a range of temperatures other than the certification temperature, these tables for DIN Buffers Standards can be viewed here.
Please note that the information carried in these tables are not certified or guaranteed, but contains typical pH values at a range at temperatures and so is indicative only.
See More on our pH Buffer Solutions / Standards DIN Value range
Restriction of Hazardous Substances (RoHS-3) Declaration European Union Directive 2015/863
All Reagecon Diagnostic products conform to the Directive (EU) 2015/863 amendment of the RoHS 3 Directive 2011/65/EU, RoHS-Recast, Article 4(1): and do not supply electrical products that contain any of the below mentioned 10 Hazardous substances above the specified limits.
SUBSTANCE:
- Cadmium (Cd): < 100 ppm
- Lead (Pb): < 1000 ppm
- Mercury (Hg): < 1000 ppm
- Hexavalent Chromium: (Cr VI) < 1000 ppm
- Polybrominated Biphenyls (PBB): < 1000 ppm
- Polybrominated Diphenyl Ethers (PBDE): < 1000 ppm
- Bis(2-Ethylhexyl) phthalate (DEHP): < 1000 ppm •
- Benzyl butyl phthalate (BBP): < 1000 ppm
- Dibutyl phthalate (DBP): < 1000 ppm
- Diisobutyl phthalate (DIBP): < 1000 ppm
Reagecon’s products are not electrical or electronic equipment as defined in the Directive referenced above. Therefore, Reagecon’s products are exempt from the RoHS directive.
Our RoHS-3 Statement can be viewed here
A2LA American Association for Laboratory Accreditation Recognitions
A2LA are a member of ILAC (International Laboratory Accreditation Cooperation).
A2LA was one of the original 28 signatories to the ILAC MRA (Mutual Recognition Arrangement) in November 2000 in Washington D.C.
This means that Reagecon’s A2LA accreditations are accepted by A2LA’s partners in over 70 countries around the world as being equivalent to the accreditations issued within their own countries.
Accreditation by an organization that is not an ILAC MRA signatory can make no such statements and there is no basis for its recognition or acceptance in other countries by other accreditors.
All ILAC MRA signatories have been rigorously evaluated by their fellow signatories to ensure their compliance with ISO/IEC 17011 (“General Requirements for Accreditation Bodies Accrediting Conformity Assessment Bodies”) and to ensure that they are implementing ISO standards within the accreditation program they offer in a manner that is consistent world-wide.
An ILAC MRA Signatory Search may be performed via the following address:
To what standard(s) are Reagecon’s Gardner Colour Reference Standards compliant with ?
Reagecon’s Gardner Colour Standards are manufactured to be compliant with the ASTM D4890-06 “Standard Test Methods for Polyurethane Raw Materials: Determination of Gardner and APHA Color of Polyols” and ISO 4630:2015 “Clear Liquids – Estimation of color by the Gardner colour scale” standards.
Temperature vs pH Value for pH Buffer Solutions (pH 4.00, pH7.00 & pH10.00 @ 20oC)
Temperature vs pH Value for pH Buffer Solutions
Reagecon’s pH Buffer Solutions / Standards have their pH value certified at a single temperature.
To facilitate use of our pH Buffer Solutions / Standards, on the bottle label, Reagecon include a table of typical pH values at a range of temperatures other than the certification temperature.
Please note that the information carried in this table is not certified or guaranteed, but contains typical pH values at a range at temperatures and so is indicative only.
pH 4.00 @ 20oC
| Temperature (oC) | pH |
| 10 | 4.05 |
| 15 | 4.04 |
| 20 | 4.00 |
| 25 | 4.00 |
| 30 | 3.98 |
| 35 | 3.98 |
| 40 | 3.98 |
| 50 | 3.96 |
| 60 | 3.96 |
pH 7.00 @ 20oC
| Temperature (oC) | pH |
| 10 | 7.04 |
| 15 | 7.02 |
| 20 | 7.00 |
| 25 | 6.98 |
| 30 | 6.97 |
| 35 | 6.96 |
| 40 | 6.95 |
| 50 | 6.95 |
| 60 | 6.95 |
| 70 | 6.95 |
| 80 | 6.94 |
| 90 | 6.97 |
pH 10.00 @ 20oC
| Temperature (oC) | pH |
| 10 | 10.18 |
| 15 | 10.12 |
| 20 | 10.06 |
| 25 | 10.01 |
| 30 | 9.97 |
| 35 | 9.93 |
| 40 | 9.89 |
| 50 | 9.83 |
pH 13.00 ± 0.05 @ 20oC
| Temperature (oC) | pH |
| 10 | 13.37 |
| 15 | 13.17 |
| 20 | 13.00 |
| 25 | 12.80 |
| 30 | 12.61 |
| 35 | 12.47 |
| 40 | 12.30 |
| 50 | 11.95 |
| 60 | 11.58 |
Analytical Volumetric Concentrates – Dilution Instructions Ampoule to make 1000ml
Analytical Volumetric Concentrates – Dilution Instructions Ampoule to make 1000ml
This ampoule allows you to obtain by dilution: 1000ml of solution.
- Put the ampoule, with the funnel upwards, into the volumetric flask and pierce the membranes by inserting the glass rod supplied.
- Pour the content into the volumetric flask and dilute to volume with distilled water.
- Carefully rinse the ampoule with distilled water.
- Ideally, this should be carried out in standard conditions e.g. 20°C
What is the shelf life of Reagecon’s Standards once opened
Reagecon Standards should be stored in their original container (in an upright position) at the applicable temperature outlined on the Product / Box Label (< – 15°C or +2 to + 8°C or +10 to +30°C), away from any strong light source (e.g. sunshine, UV lamp), heat sources and volatile chemicals. It is strongly advised to record the first opening of the bottle, to close it immediately after every use and to discard the contents of the bottle when less than 10% of the original volume of the solution is left. The expiry date as outlined on the bottle label is valid provided the product is properly stored and handled in compliance with Good Laboratory Practice, with the exception of the following parts which are intended for single use:
- Conductivity Standard 1.3 µS/cm
- Premium TOC range (single use vial)
- PAHs / Phenols / Pesticides / VOCs / PCBs / Azo Dyes / Arocolors / Toxaphene / Osmolality (in ampoules).
This expiry date ceases to be valid if the product is damaged, contaminated or modified.
What is the shelf life of Reagecon’s pH Buffer Capsules once made into a solution
Reagecon Buffer Capsules (pH 4.00, 7.00, 9.00 & pH 10.00) once reconstituted should be stored in an air tight container (in an upright position) at room temperature, away from any strong light source (e.g. sunshine, UV lamp). They will have a shelf life of 3 months provided Good Laboratory Practice is strictly adhered to, it is strongly advised to record the date of reconstitution on the bottle, to close it immediately after every use and to discard the contents of the bottle when less than 10% of the original volume of the solution is left. This 3 month expiry date is valid provided the product is properly stored and handled in compliance with these instructions. This expiry date ceases to be valid if the product is damaged, contaminated or modified.
Please note that Reagecon Buffer Capsules pH 9.00 & pH 10.00 (once reconstituted) are less stable than Reagecon Buffer Capsules pH 4.00 & pH 7.00 as these products absorb CO2 from the atmosphere, therefore it is imperative to follow the above recommendations in order to allocate a 3 month expiry date to these products.
Are Ratio and Non-Ratio turbidity standards interchangeable
No – these standards are specifically designed for use in the relevant turbidity meter and are not interchangeable.
What is the relationship between FTU and NTU units
FTU and NTU are identical. Nephelometric Turbidity Units (NTUs) are the units used to describe turbidity. Formazin was established as the first calibration standard for turbidity. The units of turbidity were originally known as Formazin Turbidity Units (FTU).
Can turbidity be measured in coloured samples
Turbidity can be measured in coloured samples provided the correct type of instrument is used. There are 2 different light sources that are used in nephelometers – infra-red (IR) light at 860nm or broad-spectrum ultraviolet-visible light (UV-Vis).
IR light (generally produced by diodes) has the advantage of being outside the visible range, and any sample coloration cannot have any interference effect. UV-Vis light (from tungsten lamps) irradiates the complete spectrum thus sample colour will affect any measurement in the range in which the coloration absorbs.
If turbidity measurements are required in a coloured sample then a nephelometers with an IR light source should be used.
How is the colour of water determined (Paltinum Cobalt, Hazen)
The Platinum-Cobalt method of measuring colour is the standard method for measuring colour. Hazen units are defined as the colour produced by 1mg platinum/litre in the form of the chloroplatinate ion. Preparation of these standards was originally described by A. Hazen, thus the term Hazen colour is also used for this method.
What kind of samples can be measured using the Hazen method (Paltinum Cobalt, Hazen)
The Hazen method is used for measuring the colour of potable water and waters in which the colour is due to naturally occurring materials. It is not applicable to most highly coloured industrial wastewaters.
Does the pH of the water sample influence its colour
The colour value of water is extremely pH-dependent and invariably increases as the pH of the water is raised. When reporting a colour value, the pH value at which the colour is determined should always be specified.
Is there any traceability for Reagecon Hazen standards
There is no primary reference standard for Hazen colour standards. The preparation and use of Hazen standards is covered in several standards, namely ISO 2211:1973, ASTM D1209 and ISO 6271. Traceability is available for the UV spectrophotometer used in testing of Hazen standards (calibrated using NIST traceable filters).
Which DPD tablets do I use to determine Chlorine in Water
There are several different types of DPD tablets available for chlorine testing, depending on what you wish to determine. Free chlorine – this is a measure of any active chlorine in solution, and is measured using DPD No.1 Total chlorine – this is a measurement of all the active and inactive chlorine compounds in water. It is measured by either using DPD No.4, or by a combination of DPD No.1 + DPD No.3 test tablets (add DPD No. 3 to a sample which has been tested for Free Chlorine). Combined chlorine – this is formed by the reaction between hypochlorous acid and ammonia / nitrogenous based compounds. It is measured by the difference between free chlorine and total chlorine tests. i.e. Total chlorine = DPD No 1. + DPD No 3 tests. In addition, there are 2 different grades of DPD tablets available, depending on the method of analysis used. Rapid grade tablets are designed for use with comparators, whereas Instrument grade tablets are designed for use with photometers. Rapid grade tablets do not dissolve fully, thus they will give erroneous readings if used in a photometer.
How can the performance of ORP (redox) Electrodes be checked
The performance of an ORP electrode can be determined either by measurement in specially prepared buffer solutions saturated with quinhydrone or with ready-to-use Reagecon Standard Redox Solution (e.g. Redox Solution 250 mV @ 25°C, Cat No. RS250). When the electrode responds properly, calibration is unnecessary. However, contaminated precious-metal electrodes can give false potential indications, and the reference-electrode potential may also be erroneous. For these reasons it is desirable to check the performance of redox electrodes, for which purpose solutions of a definite redox potential are required.
I measure the pH of samples from my effluent system and I have noticed a black deposit forming on the diaphragm of my electrode. What causes this deposit and what can I do to remove it
This deposit is formed by the reaction between sulphide ions in the effluent with silver ions from the electrode’s reference system, forming silver sulphide – an insoluble compound.
If left unchecked, the silver sulphide deposit will eventually clog the electrode’s diaphragm, reducing its accuracy and response time until eventually no meaningful readings can be taken.
The silver sulphide can be removed using Reagecon’s Inorganic Electrode Cleaning Solution (Product IECS5). The electrode should be regularly cleaned with this cleaning solution as part of a preventative maintenance program.
Further details on the care and maintenance of pH electrodes are given in the Reagecon Technical Paper “Care, Maintenance and Fault Diagnosis for pH Electrodes”
Chloride samples can interfere with COD vial reaction, do I need to take special precautions if there is chloride in my samples
Reagecon’s COD vials contain mercuric sulphate, which forms a complex with chloride ions and suppresses the chloride interference. For most samples, there is no need for additional sample preparation.
The 0 – 150mg/l and 0 – 1,500mg/l COD vials contain enough mercuric sulphate to complex up to 1,000mg/l chloride.
The 0 – 15,000mg/l vials can be used for chloride contents up to 10,000mg/l.
If possible, dilute the sample so that the chloride content is below these levels. If this is not possible, add 0.1g of mercuric sulphate for each 2,000mg/l of additional chloride. However, if a precipitate is formed then the colourimetric method will give unreliable results.
How should I use Reagecons COD Standards and what benefits will I get from using these standards give me
You should treat Reagecon’s COD standards in exactly the same way that you treat your samples for amount to add to each vial, digestion time and measuring method.
You should perform a test on a Reagecon COD standard with each batch of test samples. If an acceptable reading is obtained for the standard then you can be confident that your test sample readings will be correct; conversely, if you do not obtain an acceptable reading for standard then the test sample results will be dubious and so should be repeated.
Reagecon’s COD standards are all traceable to NIST – this means that you can claim traceability for your own readings and ensure compliance with environmental testing regulations.
I have run a COD test on a Reagecon COD standard but my result does not match the value on the Certificate of Analysis. Is this to be expected and what should I do about this difference
The methodology involved in COD testing means that there are a number of significant sources of errors. Recent years have seen improvements in instruments and testing systems; but, as is the case with all analytical measurements, COD testing is not perfect. The typical accuracy of high quality COD instruments from manufacturers such as Aqualytic is ±5% or 7.5mg/l (whichever is greatest); but other instruments may have a poorer performance. If the result for the COD standard is within the manufacturer’s accuracy specification then the result is acceptable. If this is not the case, investigation of the test method, equipment, test environment and test personnel will be required to determine the source of this unacceptable error.
I have heard that there are faster ways of measuring COD and that it can be measured with a digestion time of 30 minutes and also can be measured even faster with an online analyser. Is this true
Only an estimate of COD can be obtained with reduced digestion times. If the sample is readily oxidized then oxidation may be completed within a reduced digestion period. Systems that use a reduced digestion time do not conform to the Standard Method and so cannot be used for evaluating and reporting compliance with regulatory requirements.
The online systems do not measure COD; they measure Total Organic Carbon (TOC) and use an empirical correlation to give an indication of the COD value of the sample. This correlation will hold true if the concentration of the pollutants changes; however, if the composition of the pollutants changes then the ratio of COD to TOC will also change.
If rapid COD and online TOC measurements cannot measure the true COD value why does anyone make these measurements
The reason for using these analytical techniques is speed of analysis. Its takes at least 2½ hours to collect, prepare, digest, cool and read a true COD value; online TOC analysis can give a result in under 10 minutes and do this automatically. If there is a problem with the waste treatment then the rapid analysis allows intervention to be made quickly – e.g. incorrectly treated waste can be diverted to a holding tank rather than being discharged to a watercourse.
What are the correct storage conditions for Reagecon TOC standards (Quality Range)
Reagecon’s TOC standards (Quality Range) should be stored in their original containers, at room temperature and away from direct sunlight.
Are Reagecon TOC standards acidified (Quality Range)
No, they are not acidified, but sodium azide is added as preservative.
What is the lowest TOC standard that Reagecon produce (Quality Range)
The lowest value TOC standard that Reagecon manufactures is 5 ppm (5 mg/l). Additionally, Reagecon also offer TOC standards up to 10,000ppm.
Is there any relationship between BOD and COD
BOD tests only measure the biodegradable fraction of the total potential dissolved oxygen consumption of a water sample.
By comparison, COD tests measure the oxygen demand created by toxic organic and inorganic compounds as well as by biodegradable substances. A standard COD test, therefore, can be used to evaluate many industrial type wastes not readily analysed for water quality factors by the sewage-oriented BOD test.
Information on the wastewater biodegradability can be gained by comparing BOD and COD. A high ratio of BOD to COD shows that the wastewater is relatively biodegradable whereas a low BOD to COD ratio indicates that the wastewater is either more slowly biodegraded or contains a significant fraction of non-biodegradable material.
How do photometers measure water quality parameters
The basic principle governing colourimetric chemistry is the Beer-Lambert law. Many compounds absorb visible light and their absorbance is directly proportional to their concentration. The reagents used in colourimetric testing produce such a compound and its concentration is proportional to the parameter being examined. Hence the absorbance measured by the photometer is proportional to the concentration of the parameter under examination.
The Aqualytic PC Compact photometers use an LED as their light source. How do they work
Light Emitting Diodes (LEDs) have been chosen as the light source for the PC Compacts as they are only capable of producing light at a specific wavelength when an electrical signal is applied to them. The LED is only switched on for the duration of each test measurement. Modern LEDs are incredibly durable and can withstand many thousands of measuring cycles, giving an extremely long instrument life. High quality LEDs are inexpensive to manufacture. This means that the PC Compacts can provide high quality analysis over a long period of time at a very competitive price.
Why do I need to perform a ‘blank’ before measuring my samples in my photometer
The photometer will measure the combined absorbance of the vial (or cuvette) and the test sample. As we are only interested in the absorbance of the test sample, the absorbance of the cell needs to be subtracted from the total absorbance. This is done with a ‘blank’ vial – usually a cell filled with purified water. Most photometers will automatically subtract the blank’s absorbance from the test sample readings. For improved measurement accuracy, matched cells that are certified as having identical optical properties should be used for photometric testing.
For some test methods, e.g. COD, the unreacted test reagents have a significant absorbance at the measurement wavelength. In these instances, the blank is prepared by treating purified water with the reagents and the blank can be used to set the zero point.
How should I handle my photometer vials
It is important to handle photometer vials with care to produce accurate results. Any scratches and smudges on the vial or air bubbles within the vial will affect the measured absorbance. A lint-free soft tissue should be used to clean the outside of the vials. The vials should be stored in a foam-padded container to prevent a build up of dust and dirt. The vials should be carefully positioned in the photometer’s cell holder and the chamber lid closed to ensure that stray light does not affect the readings.
Some of my colourimetric test methods require the sample to be heated after addition of the test reagents. Why is this heating required
Some of the reactions used to produce a colour change require heating to take the reaction to completion and a vial-heating block is used for this process.
The following Aqualytic parameter tests require the use of a heater block:
- COD
- TOC
- Total Chromium
- Total Nitrogen
- Total Phosphate.
My photometer does not express results in the species that I am interested in. What can I do about this
Different countries and industries have their own preferred means of expressing water quality results, e.g. ammonical nitrogen is often expressed in terms of the N content, NH3 content or NH4+ content – all of which will have different values. Many photometers can be programmed to express the results in the species of interest. Common conversion factors are given in the table below:
What is turbidity
Turbidity is an expression of the optical property that causes light to be scattered and absorbed rather than transmitted in straight lines through a sample. Turbidity in water is caused by suspended and colloidal matter, e.g. clay, silt, fine particulate organic and inorganic matter, and microscopic organisms. The higher the concentration of such particulate matter the greater the turbidity will be.
How is turbidity measured
Turbidity is generally measured using a nephelometer, which measures turbidity in terms of the light scattered at right angles to the path of a light passing through the sample. The major components of a nephelometer are:
- A light source is a tungsten lamp operated at a colour temp of 2200K – 3000K.
- The distance traversed by the incident scattered light within the sample should not exceed 10cm.
- The detector shall be centred at 90° to the incident light not to exceed +/- 30° from 90°.
- The detector and filter system (if used) should be a clear colourless glass free form scratches & fingerprints
- The instrument sensitivity should permit detection of a turbidity difference of 0.02 unit or less in water that has a turbidity value of 1NTU or less.
These strict design guidelines have been imposed by regulatory bodies such as the EPA, ASTM and are cited in the Standard Methods. As the design criteria for instruments are broad in their acceptance limits, this accounts for variances between instruments.
What are the benefits of Reagecon’s turbidity standards (Ratio & Non Ratio) over Formazin turbidity standards
A detailed comparison of these turbidity standards is given in the Reagecon Technical Paper “Turbidity Standards and Reference Materials”, but the benefits can be summarised as:
- Non-toxic (Formazin is derived from a carcinogen).
- Shelf-life of 2 years at all values; Formazin has a shelf-life of 3 months at 4,000NTU and must be freshly prepared at low values.
- Reagecon’s turbidity standards are certified traceable to NIST Reference Materials; Formazin standards are not traceable and so do not facilitate comparability of test results.
- Reagecon’s turbidity standards have a specification of ±1%. Formazin standards are very difficult to make accurately and have typical accuracies from 10 – 30%
- Homogenity. Formazin coagulates and the particles settle out. If you shake Formazin before use, the particles collide and fragment, changing the turbidity value of the solution. Reagecon’s turbidity standards use microscopic polymer beads suspended in an emulsion and Brownian motion ensures their homogeneity.
- Certified, ready to use – no need for dilution to working values.
Can I make a lower value turbidity standard by diluting a Reagecon turbidity standard with water
You cannot dilute Reagecon’s turbidity standards with water to give a lower value standard, as the solvent for these standards is a specially formulated emulsion. However, Reagecon provide turbidity standards in a range of values covering the entire turbidity and so you can obtain a suitable, ready to use turbidity standard for all measurement applications. This means that there is no need for dilution and also means that the turbidity standard that you use on your instruments is certified and traceable to NIST Standard Reference Materials.
What is the difference between Ratio and Non-Ratio turbidity meters
The difference between these standards is due to the difference in instrument types – Non-Ratio turbidity instruments measure turbidity at 90° (+/- 30°) to the incident light (also called nephelometers), whereas Ratio instruments measure turbidity at angles other than 90°. Ratio instruments can be used for measuring the turbidity of coloured liquids.
How is acidity measured
Acidity is generally measured titrimetrically by the addition of a standard alkali solution to the sample in the presence of a suitable indicator. The acidity of the sample is calculated based on the number of drops of titrant added to give the required indicator colour change.
What is meant by the alkalinity of water
The alkalinity of water is its acid-neutralising capacity and is a significant parameter in many uses and treatments of waters and wastewaters. The alkalinity of many surface waters is primarily a function of carbonate, bicarbonate and hydroxide content, but may also include contributions from borates, phosphates, silicates and other bases if present.
How is alkalinity measured
The alkalinity of a sample is determined by titration using a standard acid (either 0.02N sulphuric or hydrochloric acid) and is expressed as equivalent milligrams CaC03 per litre. The results for alkalinity can be reported in two different units, depending on the end-point selected for the titration. p-alkalinity (phenolphthalein alkalinity) is the term commonly used for the quantity measured by titration to pH 8.3 and total alkalinity or m-alkalinity (methyl orange alkalinity) are terms commonly used for the quantity measured by titration to pH 4.5.
What is meant by the hardness of water
Total hardness is defined as the sum of the calcium and magnesium concentrations in a sample, expressed as calcium carbonate, in milligrams per litre.
How is hardness measured
Water hardness can be determined by titrating with EDTA, using Eriochrome T as indicator.
What does TDS mean
TDS stands for Total Dissolved Solids, and is a measure of the total quantity of mineral salts dissolved in water. TDS is an indicator test used for wastewater analysis, but is also used as a measure of the mineral content of bottled water and groundwater
How is TDS measured
TDS is usually measured by weight in terms of milligrams per litre (mg/L) or parts per million (ppm). It can be determined in 2 different ways, either by evaporating a filtered sample to dryness at 180°C in a weighed dish or by using a conductivity meter. TDS measurements determined by conductivity are actually a measure of the conductivity of the sample, i.e., the ability of the water to carry a charge, and not the actual total dissolved solids. This TDS value is then calculated based on the assumption that the primary dissolved minerals are either a combination of NaCl or KCl.
What is the difference between TDS and Total Solids
Total solids is the term applied to the material residue left in a vessel after evaporation of a sample and its subsequent drying in an oven at a defined temperature. Total solids include total suspended solids (the portion of total solids retained by a filter) and total dissolved solids (the portion that passes through a filter).
How is Total Solids measured
Total Solids is measured by drying a well-mixed sample in a weighed dish to a constant weight in an oven to 103 – 105°C.
How is Total Suspended Solids measured
Total Suspended Solids are determined by filtering a well-mixed sample through a weighed standard glass-fibre filter. The residue retained on the filter is then dried to a constant weight at 103 – 105°C.
I have heard that Suspended Solids can be measured online, is this correct
Online Suspended Solids instruments obviously cannot use the filtration, drying and weighing technique. Instead, they measure the turbidity of the sample stream and multiply this by a correlation factor to give a reading as a Suspended Solids value. The correlation factor depends on the composition of the sample stream. Online Suspended Solids instruments can give a very good, rapid indication of the likely Suspended Solids of the sample stream; but they cannot give a true Total Suspended Solids reading.
What is meant by BOD
BOD stands for Biological Oxygen Demand, and represents the amount of oxygen consumed during the aerobic digestion of organic matter by the microorganisms present in a sample of water.
Why is BOD important
BOD is an important measurement for many industrial water samples. Although it is an empirical test, it is used to determine the relative oxygen requirements of wastewaters, effluents and polluted waters.
How is BOD measured
BOD can be measured by three different techniques – the Winkler method, by using a dissolved oxygen probe or manometrically.
The Winkler method is a titrimetric wet chemistry test that measures the amount of oxygen present based on conversion of oxygen to iodine. Although considered by some to be it the “Gold Standard” in BOD determination, it requires skilled and time-consuming sample preparation; is laborious to perform and so is relatively expensive.
The dissolved oxygen (DO) method of BOD determination involves the electrochemical measurement of DO in the sample.
The manometric method. The sample is placed in an airtight bottle, with a pressure sensor mounted on the bottle. The carbon dioxide evolved during aerobic respiration is absorbed using an alkali (KOH). As oxygen is used up during aerobic respiration and the carbon dioxide is absorbed there will be a pressure drop in the bottle proportional to the amount of oxygen used. This pressure drop is measured and the oxygen consumption (BOD) is calculated from the pressure drop. The manometric method requires less sample preparation and less reagents than the other 2 methods. Manometric measuring instruments are also able to take readings automatically and store them for retrieval at a later time.
BOD levels are expressed in terms of mg/l O2 and are measured over a 5-day period. The samples must be maintained at 20°C ±1°C for the duration of the test.
What is the purpose of adding Nitrification Inhibitor to BOD samples
BOD is defined as the amount of oxygen required by microorganisms to digest organic carbon. However, there are bacteria that use oxygen to convert NH4+ to NO2- and NO2- to NO3- by a process called nitrification. Adding Nitrification Inhibitor (N-Allylthiourea) to the samples, prevents the nitrification process so that only oxygen demand due to carbon digestion is measured.
What sample preparation is required for the manometric BOD test method
Using Aqualytic’s OxiDirect BOD System, there is very little preparation required for most. The process for starting a test is:
- Depending on the required measuring range, fill the appropriate overflow flask with the sample and transfer this to a BOD bottle.
- Add the appropriate number of drops of nitrification inhibitor and a magnetic stirrer to the sample.
- Add 3 drops of 45% potassium hydroxide to the rubber seal gasket. Place the gasket in the neck of the bottle and screw the BOD sensor head onto the bottle.
- Place the BOD bottle on the OxiDirect base and start the measurement process, including inputting the sample size.
What is meant by COD
Chemical Oxygen Demand, COD, is a quantitative measure of the amount of oxygen required to oxidize all organic components in a sample of waste water, both non-biodegradable and biodegradable. It is used as a rapid measure of organic pollutants in water.
How is COD measured
COD is generally measured by the closed reflux colorimetric method (Standard Methods 5520D). It involves the chemical oxidation of any organic matter in the sample using potassium dichromate in a 50% sulphuric acid solution at 150°C for 2 hours.
What sample volumes are used for Reagecon COD vials
The Reagecon COD vials are equivalent to the Hach range of COD vials and use the same sample sizes:
- 2ml for 0 -150 and 0 – 1500 mg/l vials
- 0.2ml for 0 – 15,000mg/l vials
Comparative Study of the Performance of Reagecon COD vials and Hach COD vials
The subject of the above question is comprehensively covered in the technical paper entitled A comparative study of the performance of Reagecon COD vials and Hach® COD vials using the Hach® DR/2010 photometer published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
What practical steps should be performed when calibrating a pH meter
When carrying out the calibration the following points should all be borne in mind
- Electrodes should never be allowed to run dry because this will create static charges on the glass membrane resulting in a very sluggish response.
- Cleaning of the electrode between buffers and sample or from sample to sample should be carried out by a jet of water from a wash bottle, followed by careful rinsing with the buffer or sample to be measured. For very accurate work it is recommended to repeat the zero calibration after slope adjustment.
What do I need to do to check that my pH meter is functioning correctly
To have full confidence in your pH measurements it is essential that you can demonstrate that each component of the measuring system is functioning to specification. If any component of the measurement system does not meet this requirement, questionable results will be obtained. For pH measurement, the following must be verified:·
- The pH meters ability to correctly process both pH and temperature inputs correctly
- The pH electrodes ability to generate the correct output signal
- The temperature sensors ability to generate the correct output signal
Routine calibration using pH buffers will not demonstrate all of the above, this is why annual calibration should be performed.
What does annual calibration involve
To fully demonstrate that all components of the pH measuring system are functioning correctly, the following steps should be performed:
- Certified, traceable electrical simulators and standards should be used to verify that the pH meter is processing input signals correctly.
- The performance of the temperature probes should be compared to a certified reference thermometer.
- The pH meter and electrode should be calibrated using certified, traceable pH buffers.
- The correct performance of the pH electrode should be verified using certified, traceable control standards.
For most pH users it is not practical to purchase and maintain a certified traceable pH simulator, as it is expensive. Reagecon’s Technical Services Department can provide a calibration service to customers that meets all their requirements, be it Instrument Calibration or Validation.
What is the difference between Instrument Calibration and Validation, and which one do I need
It is a regulatory requirement for pharmaceutical companies to perform Instrument Validation on new equipment, and regular Requalification on existing equipment. In brief, Instrument Validation is a fully documented process that ensures the instrument is working correctly.
It demonstrates that the measuring instrument is working correctly and hence has no detrimental effect on the quality of the analytical results. For industries which are not as heavily regulated annual calibration (as previously described) would be sufficient to demonstrate that the instrument is working correctly.
Reagecon’s Technical Services Department has developed calibration and validation protocols for a wide range of pH instruments. This service can be performed at the customers’ premises, or alternatively the instrument can be sent to Reagecon for calibration in our purpose-built Metrology Laboratory.
My pH electrode has a hole with a detachable cover at the top of the glass section. What is the purpose of this hole
This hole serves two purposes – to allow replacement of the electrolyte and also to allow pressure to be equalised during measurement. If the hole is covered during measurement then the electrolyte will not be able to travel through the electrode’s diaphragm into the sample thus the electrode accuracy and responsiveness will be reduced.
Some electrodes have a hole at the top for replacing the electrolyte, but others dont. Why is this
This electrode contains the electrolyte in a gel format. It is non-refillable, and thus is low maintenance. Gel filled electrodes are less accurate than liquid filled electrodes due to a slower response resulting from liquid potentials. In addition, if the electrolyte becomes poisoned it cannot be changed, and the electrode must be replaced.
What applications should gel filled electrodes be used for
Gel filled electrodes are generally supplied with plastic bodies. They are thus more rugged than liquid filled glass electrodes, hence they are commonly used with portable meters.
Measuring the pH of water from a water purification system can be problematic. Why is this
The objective of a water purification system is to produce a finished product of low ionic strength. This also means that there is low hydrogen ion concentration present, which can result in slow response times and inaccurate pH results if using a general-purpose pH electrode. The problem can be overcome by selecting an electrode that has the following characteristics:
- A diaphragm with a high outflow rate of electrolyte.
- An A glass membrane (gives a short response time in low ionic strength media).
The Schott Blueline 13 electrode combines both of these features in a laboratory electrode and is thus ideal for this application.
What is the alkaline error in pH measurement
In strongly alkaline solutions measurement errors can appear due to a contribution to the potential from alkali ions – Lithium (Li+) and Sodium (Na+) ions in addition to that of the hydrogen ions. Na+ and Li+ ions interact with the glass membrane and can partially or completely replace the H+ ions present. The resulting low hydrogen ion activity results in the observed pH reading being more basic than the true value. This effect occurs in solutions with a pH value > 11 and increases with increasing temperature. The magnitude of the alkali error depends on the concentration of the interfering ion with respect to the H+ ion concentration. Membrane glasses with large alkali errors have only a short service life in the presence of high concentrations of alkali metal ions
How can the pH of a strong alkaline solution be measured accurately
If you need to perform measurements in solutions containing high concentrations of alkali metal ions it is advisable to use a special membrane glass type (e.g. Schott membrane type H used in the H62 electrode) when the pH value exceeds pH 11.
How can the pH of a non-aqueous liquid be measured
The pH scale is defined in terms of hydrogen ion activity in aqueous solution and is not therefore strictly applicable to pH measurement in non-aqueous systems. If measurements are to be performed in non-aqueous liquids, there are several problems which may be encountered. The low ionic strength and low conductivity of some non-aqueous solvents may result in high noise or drift. In addition, the glass membrane of the pH electrode may become dehydrated from immersion in the non-aqueous solvent and thus lose its ability to respond to H+ ions.
The ideal way to minimize these effects is to use a separate reference and measuring electrode with an auxiliary salt bridge containing a non-aqueous electrolyte filling solution. If this is not available a standard pH electrode may be used providing the following steps are performed:
- Select an electrode with a low resistance membrane and a free flowing diaphragm such as the Schott Blueline 13.
- Use an electrode containing a non-aqueous filling solution (such as saturated Lithium Chloride in Ethanol for non-polar solvents or Lithium Chloride in Glacial Acetic Acid for polar solvents).
- Soak the electrode in the non-aqueous solvent for ten minutes after standardisation and before use in the sample.
- Between successive measurements, the electrode should be rinsed with the non-aqueous solvent used to dissolve the sample.
If the reading begins to drift after considerable time in a non-aqueous solvent, the pH glass bulb must be rehydrated by immersion in an aqueous buffer.
What working life can I expect from a pH electrode
The working life of a pH electrode is heavily dependant on the application, and how it is maintained. A well-maintained pH electrode will function for 6 to 24 months in most typical applications. However, the life span of an electrode will be greatly reduced by exposure to high temperatures, high process pressure, or by exposure to aggressive media (such as HF or very caustic applications) which can result in damage to the membrane or reference system. As the membrane on a pH electrode ages the speed of response reduces decreases until it becomes unacceptably slow.
How should I store my pH electrodes when not in use
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
Why should I store electrodes in Electrode Storage Solution
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
How should I clean my pH electrode
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
How should I clean my electrode between pH measurements
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
How can pH electrodes be rejuvenated
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
What does the term drift mean in relation to pH measurement, and why should I be concerned about it
The subject of the above question is comprehensively covered in the technical paper entitled Care, Maintenance and Fault Diagnosis for pH Electrodes published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team.
The technical paper includes details of practical remedial actions which can be taken to improve the accuracy of pH measurements and extend the working life of pH electrodes.
What is the minimum level of filling solution to use in a refillable pH electrode
The filling solution level must be higher than the sample level to maintain a uniform flow of filling solution. A level of at least 2cm above sample height is recommended.
What is meant by the acidity of water
The acidity of water is its quantitative capacity to react with a strong base to a designated pH value. It is important because acids lead to corrosiveness and influence chemical reaction rates and biological processes. Its measurement can also reflect a change in the quality of the source water.
How does a pH meter measure the response of a pH electrode
The potential difference developed between the reference electrode and sensing electrode is dependant on the activity of the hydrogen ions in solution. For an ideal electrode the response is defined by the Nernst equation:
E = Eo – 2.3 (RT/nF) log aH+
where:
E = total potential (in mV) developed between the sensing and reference electrode
Eo = standard potential of the electrode at aH+ = 1 mol/l
R = Gas constant
T = temperature °K
n = valency of ion
F = Faraday constant
aH+ = activity of the hydrogen ion in solution
The term 2.3RT/nF is referred to as the Nernst slope. For an ideal electrode the slope at 25°C is 59.16 mV per decade change in hydrogen ion activity (see Figure 1).
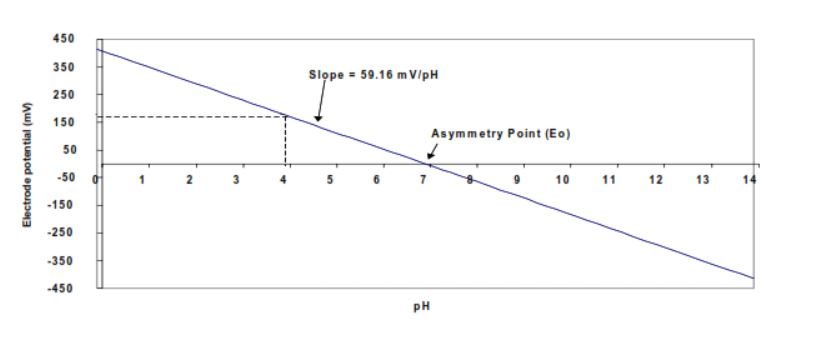
Figure 1 Slope of an ideal electrode @ 25°C
In practice the response of a real electrode does not exactly follow that predicted by the Nernst equation and must be compensated for. During calibration the meter determines the actual slope and asymmetry potential for the electrode in use and then matches the pH meter to the correct characteristics of the electrode in use.
What does the term asymmetry potential mean
For an ideal electrode the potential difference between the reference and measuring electrode is 0mV at pH 7. As the ideal electrode doesn’t exist the deviation of a few mV at pH 7 that is found with real electrodes is called the asymmetry potential, Eo. In practical terms it is a measure of the performance of the reference electrode – if the result is outside of +/- 15mV this is indicative of problems with the reference electrode such as contaminated electrolyte or reference element poisoning.
What are the acceptable ranges for the slope and asymmetry potential of a pH electrode
For an ideal electrode the theoretical slope is -59.16 mV/pH and the asymmetry potential at pH 7 is 0 mV. In practice, a range of -55 mV/pH to -60 mV/pH (or 95 102%) and an asymmetry potential of +/- 15 mV is acceptable.
What is a combination electrode
All pH sensors consist of a measuring glass electrode and reference electrode. A combination electrode is an electrode which contains both the reference and measuring electrode in the one body.
What is the function of the reference electrode
The reference electrode provides a constant potential regardless of the composition of the solution it is placed in. The voltage difference between the reference and measuring electrodes is a function of the pH value of a solution. Common reference systems in use are Silver/Silver chloride/ (Ag/AgCl) and Mercury/Calomel (Hg2Cl2). The Silver/Silver Chloride reference is far the most frequently employed because it has universal applicability, a wide operating temperature range and is relatively non-toxic.
What does the term resistance mean in relation to pH electrodes, and why is it important
This is a measure of the electrical resistance of the measuring electrode of a pH electrode. It is influenced by the chemical composition of the glass, its thickness and shape. The resistance of pH membrane glass is important as it increases with decreasing temperature. A change in resistance leads to a change in electrode response, in particular the higher the resistance of the membrane the slower the electrode response is. This can result in an electrode not being useable at a particular temperature. For example, electrodes designed for low alkaline error at high temperatures are generally of higher than normal resistance and should not be used at all at low temperatures. For this reason manufacturers specify temperature ranges in which their electrodes can be used with confidence.
What exactly is the function of the diaphragm on a pH electrode, and why is it important
The diaphragm on a pH electrode allows a flow of electrolyte from the reference solution into the solution being measured.
This completes the electrical circuit between the reference and sensing electrode and gives the potential difference which is measured by the pH meter. The type of diaphragm used will influence the outflow of electrolyte into the sample, which will in turn effect the response speed of the electrode. The common diaphragm types are listed below along with their inherent properties. For most common applications a platinum diaphragm is the one of choice.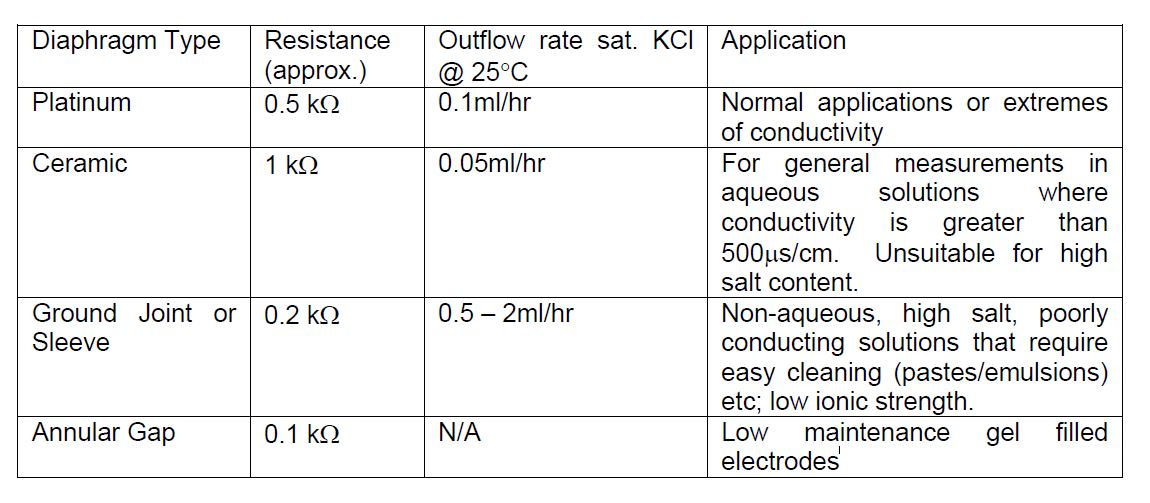
Why does the glass bulb at the bottom of the electrodes come in a variety of shapes
The glass membrane on pH electrodes comes in a variety of different shapes based on the fitness for purpose of the electrode. The most common membrane shapes in use today are outlined below:
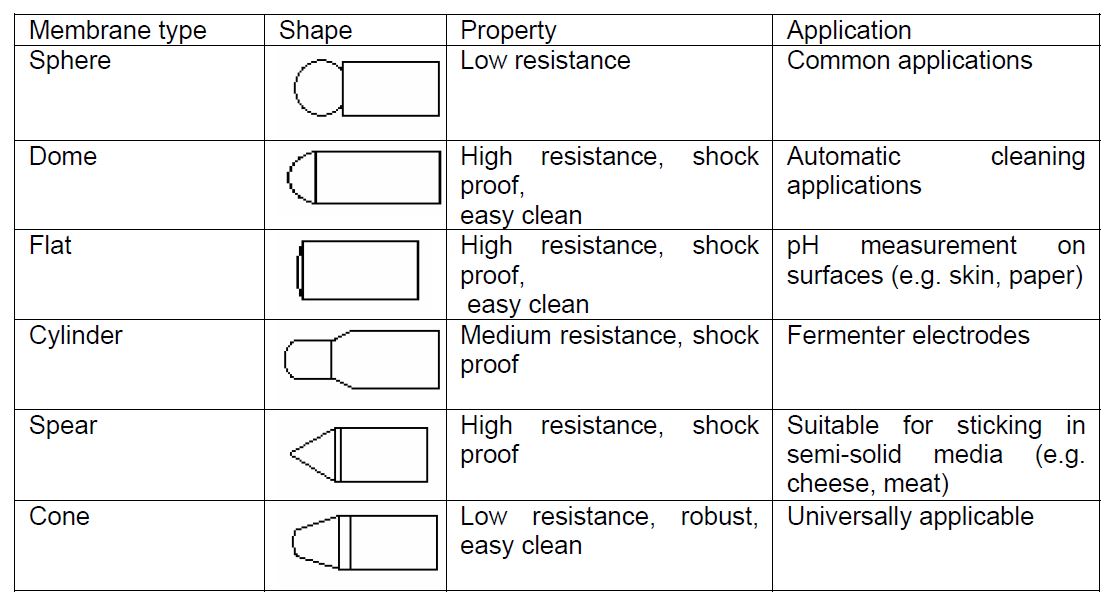
In practice there is a trade-off between the speed of response of the electrode and its ruggedness, thus the application the electrode is being selected for will govern the choice of membrane design. The classical sphere shaped membrane offers low electrical resistance due to its large surface area but is relatively fragile. The cone shaped membrane is seen as a universal design as it is robust, of medium resistance and is easy to clean.
What is the difference between the various types of membrane glass for pH electrodes
As pH is measured in a wide variety of solutions at varying temperatures it is not possible to formulate a single pH membrane glass which can give accurate and reliable pH measurements in all conditions. For this reason different types of membrane glass have been developed to give good performance in a variety of measurement conditions. The most common membrane glass types found in Schott electrodes are as outlined below:

If the wrong membrane type is selected for a particular application it may be rendered unusable without any obvious signs of change.
Does temperature have any effect on pH measurement
The subject of the above question is comprehensively covered in the technical paper entitled The Effects of Temperature on pH Measurement published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team. It includes details of practical remedial actions which can be taken to reduce or eliminate these effects.
What practical steps can I take to reduce the effects of temperature on pH measurement
The subject of the above question is comprehensively covered in the technical paper entitled The Effects of Temperature on pH Measurement published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team. It includes details of practical remedial actions which can be taken to reduce or eliminate these effects.
Does Automatic Temperature Compensation (ATC) compensates for sample measurements taken at different temperatures
The subject of the above question is comprehensively covered in the technical paper entitled The Effects of Temperature on pH Measurement published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team. It includes details of practical remedial actions which can be taken to reduce or eliminate these effects.
What does ATC Automatic Temperature Compensation actually do
The subject of the above question is comprehensively covered in the technical paper entitled The Effects of Temperature on pH Measurement published by J. J. Barron, C. Ashton. & L. Geary of the Reagecon Technical Services team. It includes details of practical remedial actions which can be taken to reduce or eliminate these effects.
What does the term junction potential mean
This is the portion of the total observed potential difference between the measuring and reference electrode that is formed at the liquid / liquid junction between the reference electrode filling solution and the test sample. The contribution of the liquid junction potential to the measured potential should be kept as small and as constant as possible. This can be achieved by careful selection of electrode, and good probe maintenance.
What affect does junction potential have on pH readings
The flow rate of electrolyte from the reference electrode into the sample is affected by the design of the electrode diaphragm. Low or irregular flow rates may cause variation in the liquid junction potential which will result in erroneous readings. This is why it is important to select the correct electrode for the application of interest.
What is meant by the response time of an electrode, and why is it important
The response time of an electrode is the length of time necessary to get a stable reading when the electrode is moved from one solution to another of different pH or temperature. Response time is dependent on the electrode type, the measuring sample, temperature, the magnitude and direction of concentration change and the presence of interfering substances. A slow response time can be indicative of the incorrect selection of electrode for the intended application.
There is a large number of different types of pH buffers, what is the difference between them and what are they used for
Reagecon manufactures the most comprehensive range of pH buffers which are designed to suit all end user requirements.
The most popular pH buffer types manufactured are outlined below:
| Buffer Type | Description |
| pH buffer @ 20°C | Value assigned at 20°C to ±0.01 pH units |
| pH buffer @ 25°C | Value assigned at 25°C to ±0.01 pH units |
| Technical Buffers | Manufactured in accordance with DIN 19266 and tested at 25°C to ±0.02 pH units |
| Professional Range | Value assigned at 20°C as per N.I.S.T and at 25°C as per DIN 19266 |
| Buffer capsules | Value assigned at 25°C to ±0.02 pH units |
All of these buffers are manufactured to exacting specifications and are supplied with detailed Certificates of Analysis which shows traceability to a N.I.S.T. SRM.
In addition to the type of buffer, different pack options are available to suit all needs:
| Pack Options | Uses/Advantages |
| 500ml, 1L & 5L | Suitable for wide range of uses |
| Colour Coded Buffers | Ease of identification |
| Twin Neck Bottle | Ideal for field work as there is no need for a separate measuring container. Also avoids possibility of contaminating bulk solution |
| Bag In Box | Available in 5L or 10L size, ideal for large volume users |
| Buffer Capsules | Ideal for users who perform calibration infrequently or where storage is an |
Does it make any difference which pH buffer I use to calibrate my pH meter?
Most modern pH meters are supplied with the facility for automatic buffer recognition. The manufacturer will have programmed in a pH versus temperature profile into memory for a specific type of buffer such as DIN or NIST (see table below)
| DIN 19266 Buffers @25°C | NIST Buffers @ 20°C |
| 1.679 ± 0.010 | 1.677 ± 0.010 |
| 3.776 ± 0.010 | 3.788 ± 0.010 |
| 4.005 ± 0.010 | 4.001 ± 0.010 |
| 6.865 ± .0.010 | 6.879 ± 0.010 |
| 7.413 ± 0.010 | 7.429 ± 0.010 |
| 9.180 ± 0.010 | 9.225 ± 0.010 |
| 10.012 ± 0.010 | 10.062 ± 0.010 |
| 12.454 ± 0.050 | 12.627 ± 0.050 |
If a different buffer to that specified by the manufacturer is used for calibration this will result in calibration errors. If you have any doubt on the best buffers to use for your pH meter then the Reagecon Technical Services team will be happy to advise you.
Should I stir my sample when taking a pH measurement
It is not strictly necessary to stir samples for pH measurement. Although stirring the solution ensures a quicker response, if the buffer solutions are stirred, then the sample should also be stirred. It is important that the stirring is carried out at the same speed for both samples and buffers. By stirring at a uniform rate this ensures a representative sample measurement is obtained (this is particularly important for colloidal or particulate samples) and also improves electrode response time. Electrodes with ceramic diaphragms suffer from interference under stirring. The platinum diaphragm on the other hand does not suffer from this interference and is thus the diaphragm of choice for most common applications.
What value pH buffers should I use to calibrate my pH meter
For proper calibration, buffer solutions of carefully selected accuracy and pH range are required. The calibration should be performed using two buffers whose values bracket the expected value of the samples being measured. In practice many pH meters are equipped with auto buffer recognition – this requires the use of the buffer values specified by the meter manufacturer. It is essential that the difference between the two calibration buffers should be no more than 2 – 3 pH units apart. This represents 2 – 3 decades of difference in pH value (i.e. a 100 to 1,000 times increase in H+ ion concentration).
Why should control standards be used when measuring pH?
For confidence in pH measurement control standards should be run regularly as a check on the functioning of the measurement system. If a control sample is run after calibration and the expected result obtained this will give the user a 99% confidence level in the calibration of the meter, and any results obtained for subsequent sample measurements. In a situation where multiple users are performing pH measurements the use of a control sample will provide confidence in the results being obtained from the different operators. The control buffer selected for use should be within the calibration range of the meter.
How are pH values measured?
pH is a potentiometric measurement where the electrode used produces an electrical signal which the pH meter converts to a pH reading. The signal produced and measured is a voltage and since voltage is a potential difference, two potentials are needed to make a measurement:
- The sensing electrode provides a potential proportional to the logarithm of the hydrogen ion activity in the sample.
- The reference electrode ideally provides a stable and consistent potential independent of the activity of the sample.
The potential difference between the reference and sensing electrode is measured by the meter and converted into a pH value.
What is the exact meaning of the term pH ?
pH as a term was coined by Sorensen in 1909. He described the concentration of hydrogen ion as a logarithmic scale which he called “Pondus Hydrogeni” or the exponent of hydrogen. pH is a quantitative unit of measure which describes the degree of acidity or alkalinity of a substance. It is measured on a scale of 0 to 14. The term pH is derived from “p” the mathematical symbol of the negative logarithm, and “H” the chemical symbol of Hydrogen.
The formal definition of pH is the negative logarithm of the hydrogen ion concentration i.e. pH = -log10[H]+.
In practice it is the hydrogen ion activity which is measured rather than its concentration.
My conductivity meter has a temperature compensation option called ‘Non-linear function’. What does this mean and should I use it for my measurements ?
Many better quality conductivity meters are programmed with this option for environmental testing. If the conductivity meter is programmed with a non-linear temperature compensation function that conforms to ISO 7888, this type of temperature compensation will give a reasonable estimate of what your sample’s conductivity value would be at 25ºC. There are strict conditions for applying ISO 7888 temperature compensation – the sample conductivity must be between 60 – 1,000 µS/cm and the measurement temperature must be between 0 – 35ºC.
More detailed technical information on the effects of temperature on conductivity measurement is given in our Technical Paper: The Effect of Temperature on Conductivity Measurement.
Specific product information can be obtained at by visiting Reagecon’s Webshop: Conductivity Standards
